Hydra CE Study Results
One-year Follow-up Study Results ( N=157) 1
Safety Endpoints
| Variables | At 1 month | At 1 year* | ||
|---|---|---|---|---|
| Events | 95% CI | Events | 95% CI | |
| All-cause mortality | 11 (7.0) | 3.7, 12.5 | 23 (14.6) | 9.7, 21.4 |
| Cardiovascular mortality | 9 (5.7) | 2.8, 10.9 | 13 (8.3) | 4.7, 14.0 |
| Non-cardiovascular mortality | 2 (1.3) | 0.2, 5.0 | 10 (6.4) | 3.3, 11.7 |
| Device-related mortality | 5 (3.2) | 1.2, 7.7 | 5 (3.2) | 1.2, 7.7 |
| All stroke | 1 (0.6) | 0.03, 4.04 | 1 (0.6) | 0.03, 4.04 |
| Disabling | 1 (0.6) | 0.03, 4.04 | 1 (0.6) | 0.03, 4.04 |
| Nondisabling/Transient ischemic attack | 0 (0) | - | 0 (0) | - |
| Life-threatening bleeding | 3 (1.9) | 0.5, 5.9 | 3 (1.9) | 0.5, 5.9 |
| Major bleeding | 7 (4.5) | 2.0, 9.3 | 7 (4.5) | 2.0, 9.3 |
| Major vascular complication | 7 (4.5) | 2.0, 9.3 | 7 (4.5) | 2.0, 9.3 |
| Acute kidney injury | 1 (0.6) | 0.03, 4.04 | 1 (0.6) | 0.03, 4.04 |
| Myocardial infarction | 0 (0) | - | 0 (0) | - |
| New permanent pacemaker implantation | 17 (10.8) | 6.6, 17.0 | 18 (11.5) | 7.1, 17.8 |
| In pacemaker naïve patients | 17 (11.7) | 7.2, 18.4 | 18 (12.4) | 7.7, 19.2 |
Data expressed as frequency (percentages). *Based on Kaplan-Meier estimate.
Performance Outcomes
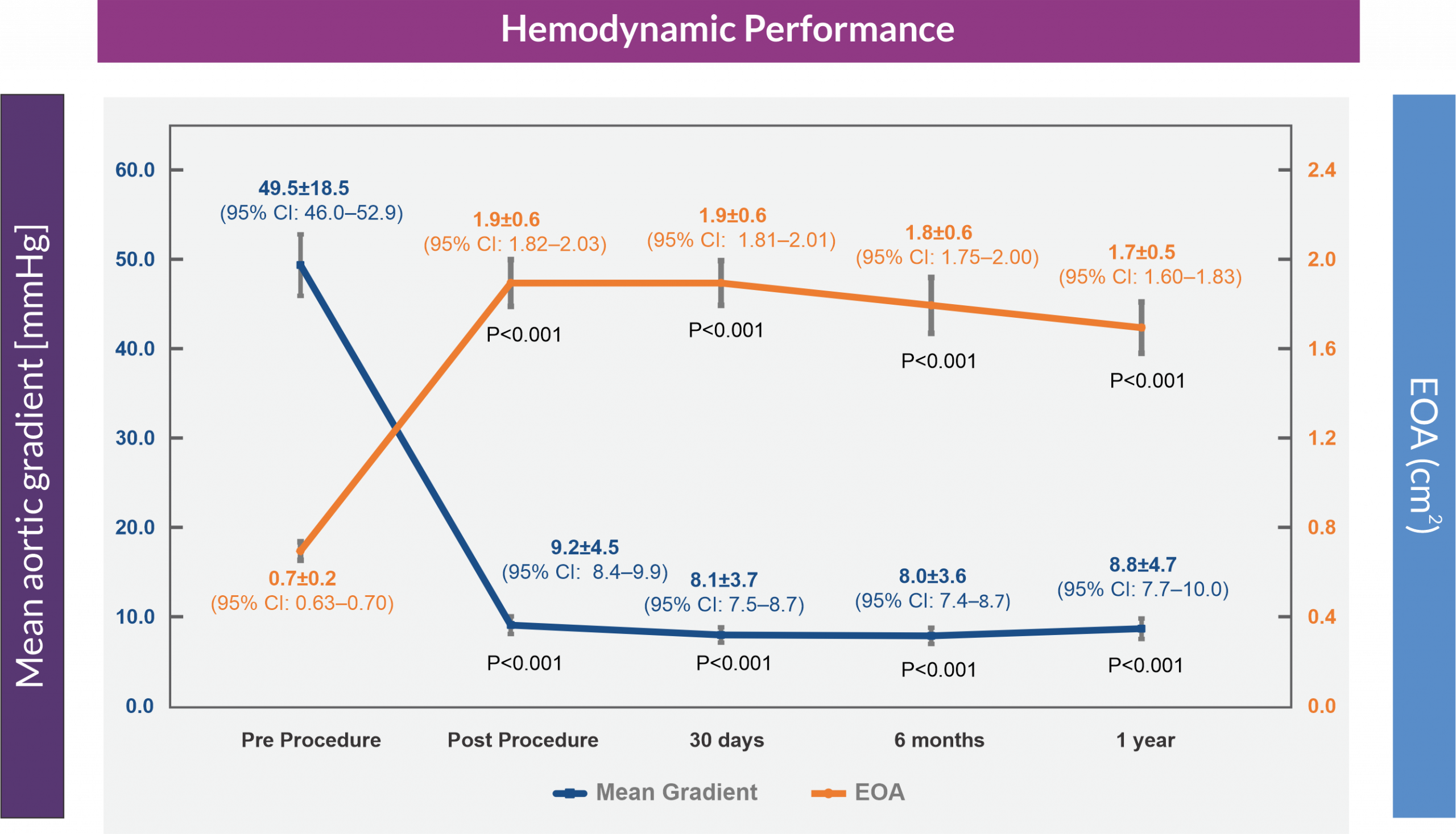
Mean Aortic Valve Gradient and Effective Orifice Area (EOA)
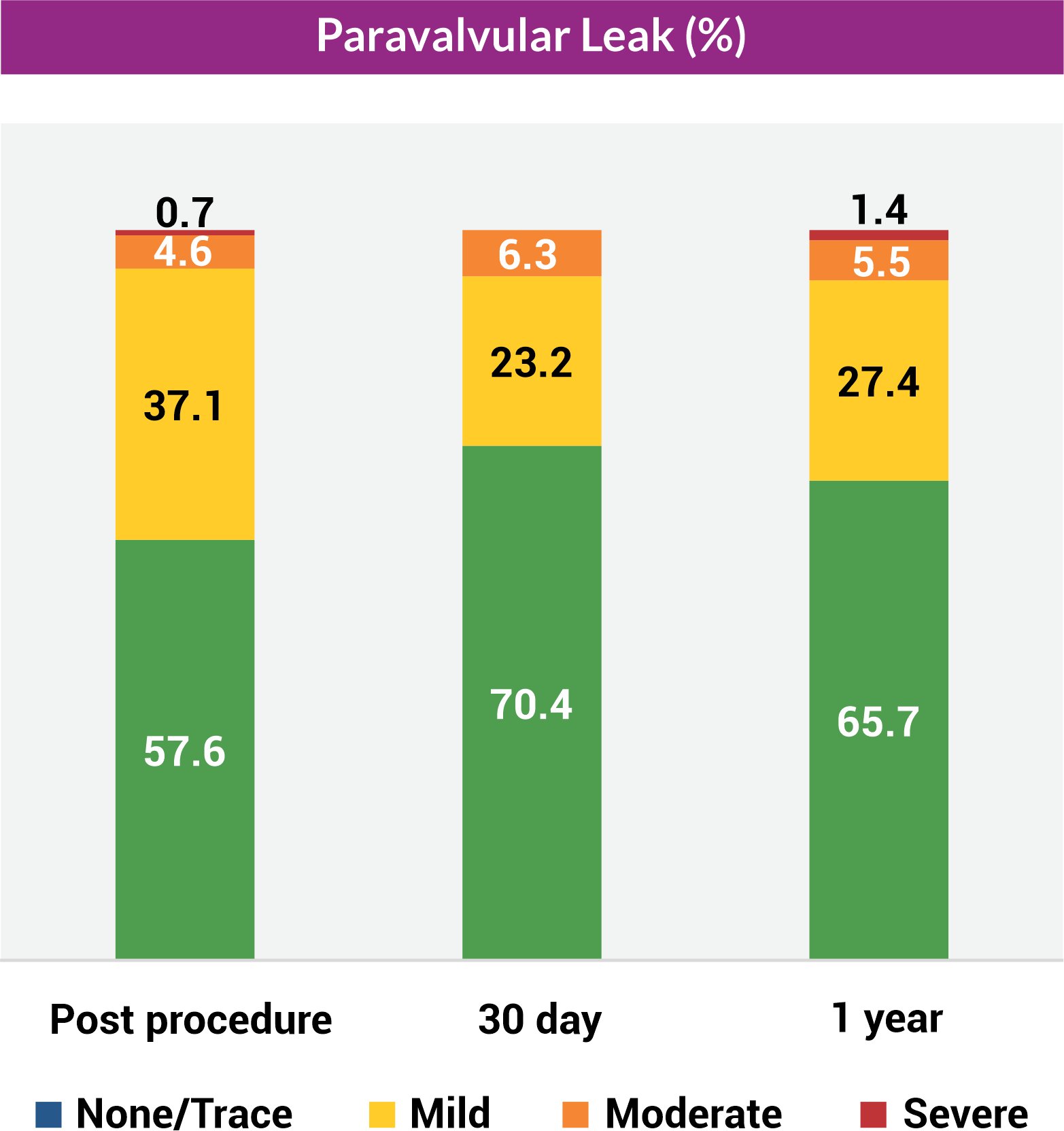
Paravalvular leak
Conclusion
The results of the Hydra CE study demonstrated favorable and sustained 1-year efficacy providing large effective orifice area and low transvalvular gradient as well as high survival rates and acceptable complication rates with regard to paravalvular leak, new permanent pacemaker, stroke, major vascular complications, and bleeding with the self expanding Hydra THV in patients with symptomatic severe AS and high or extreme surgical risk.
Long-term Follow-up Study Results (N=54) 2
Safety Endpoints
| Clinical Outcomes (N=54) | 30 days | 1 year | 3 years | 5 years |
|---|---|---|---|---|
| All-cause mortality, n (%) | 1 (1.9) | 4 (7.4) | 8 (11.8) | 13 (24.3) |
| Cardiovascular mortality | 1 (1.9) | 1 (1.9) | 2 (3.7) | 3 (6.0) |
| Non-cardiovascular mortality | 0 (0) | 3 (5.6) | 6 (11.1) | 10 (19.4) |
| All stroke | 0 (0.0) | 0 (0.0) | 4 (7.4) | 6 (12.2) |
| Disabling stroke | 0 (0.0) | 0 (0.0) | 1 (1.9) | 2 (4.6) |
| Nondisabling/Transient ischemic attack | 0 (0.0) | 0 (0.0) | 3 (5.6) | 4 (8.0) |
| Life-threatening bleeding | 1 (1.9) | 1 (1.9) | 1 (1.9) | 1 (1.9) |
| Major bleeding | 1 (1.9) | 1 (1.9) | 1 (1.9) | 1 (1.9) |
| Major vascular complication | 2 (3.7) | 2 (3.7) | 2 (3.7) | 2 (3.7) |
| Acute kidney injury | 1 (1.9) | 1 (1.9) | 1 (1.9) | 1 (1.9) |
| Myocardial infarction | 0 (0) | 0 (0) | 1 (1.9) | 3 (5.6) |
| New permanent pacemaker implantation | 7 (12.9) | 7 (12.9) | 7 (12.9) | 7 (16.7) |
| In pacemaker naïve patients | 7 (15.2) | 7 (15.2) | 7 (15.2) | 9 (19.6) |
Data expressed as frequency (percentages).
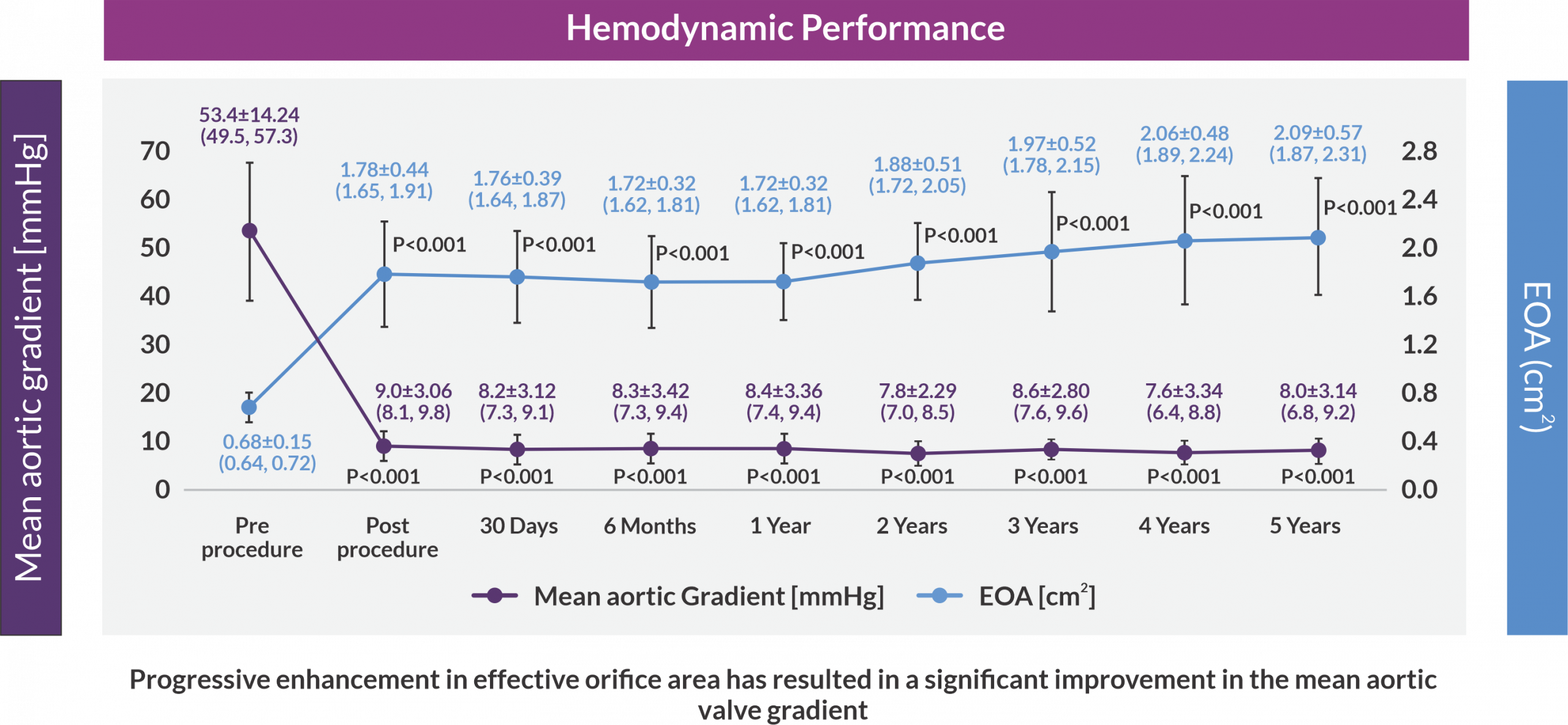
Mean Aortic Valve Gradient and Effective Orifice Area (EOA)
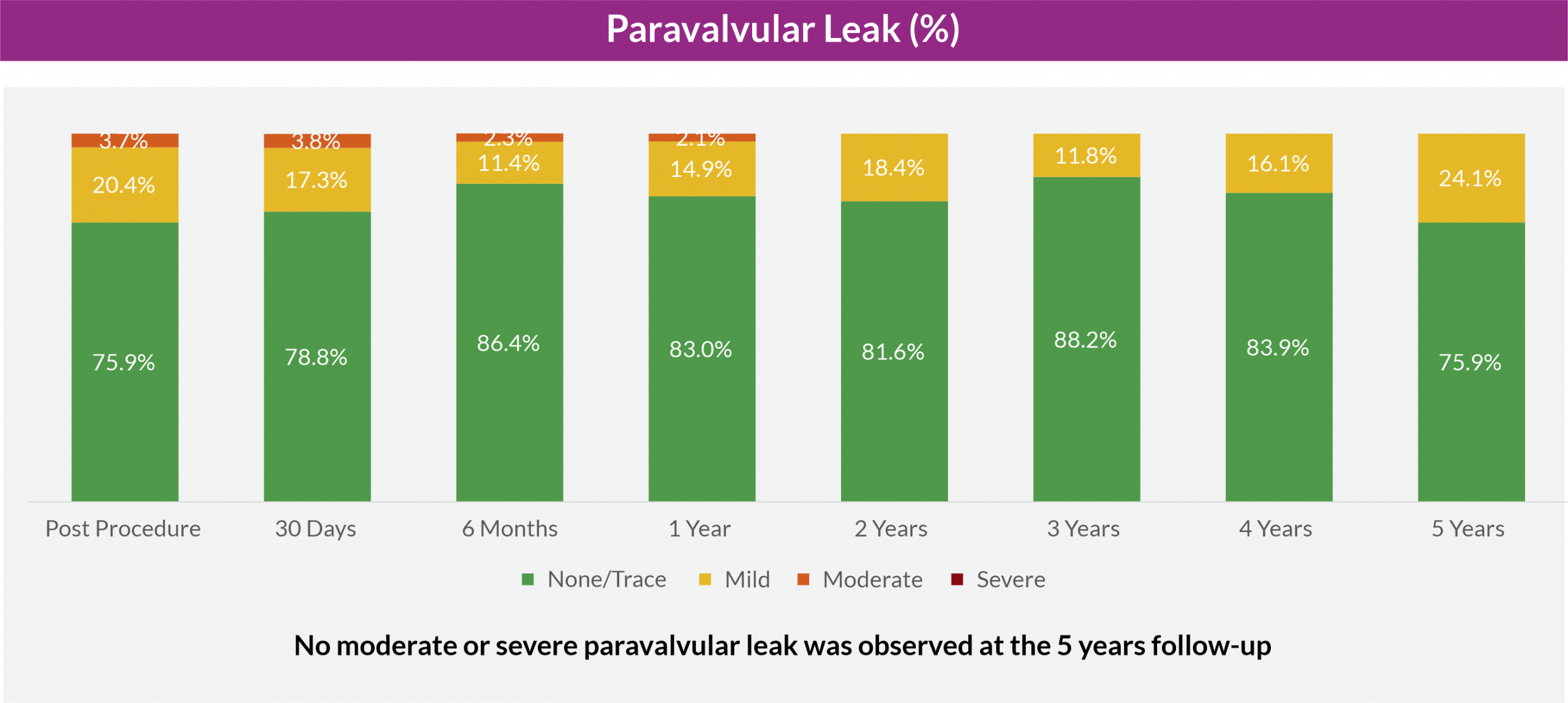
Paravalvular leak
Conclusion
The 5-year results from the long-term follow-up cohort of the Hydra CE study established excellent valve durability and sustained hemodynamic performance. The study demonstrated favorable long-term safety and effectiveness or clinical performance of Hydra THV, with low cardiovascular mortality accounting for acceptable survival rates, and low rates of disabling stroke at 5-year follow-up.
Additional Information
Baseline Characteristics
| Characteristics | One-year Follow-up Cohort (N=157) | Long-term Follow-up Cohort (N=54) |
|---|---|---|
| Age (years) | 79.2±7.1 | 81.0 ± 4.1 |
| Age <75 years | 32 (20.4) | 3 (5.6) |
| Male | 65 (41.4) | 15 (27.8) |
| Body-mass index | 26.0±5.2 | 28.6 ± 5.3 |
| Body surface area, kg/m2 | 1.7±0.2 | 1.8 ± 0.2 |
| Coronary artery disease | 65 (41.4) | 21 (38.9) |
| Previous CABG | 22 (14.0) | 9 (16.7) |
| Previous PCI | 38 (24.2) | 12 (22.2) |
| History of MI ≥6 months | 17 (10.8) | 9 (16.7) |
| History of atrial fibrillation | 30 (19.1) | 18 (33.3) |
| Left bundle branch block | 7 (4.5) | 5 (9.3) |
| Right bundle branch block | 10 (6.4) | 7 (13.0) |
| Permanent pacemaker | 12 (7.6) | 8 (14.8) |
| Heart Failure | 14 (8.9) | 8 (14.8) |
| NYHA Functional Class | ||
| Class-I | 1 (0.6) | 0 |
| Class-II | 77 (49.0) | 16 (29.6) |
| Class-III | 72 (45.9) | 38 (70.4) |
| Class-IV | 7 (4.5) | 0 |
| Diabetes mellitus | 48 (30.6) | 11 (20.4) |
| Hypertension | 125 (79.6) | 51 (94.4) |
| Prior stroke | 13 (8.3) | 3 (5.6) |
| Prior TIA | 0 | 0 |
| Peripheral artery disease | 4 (2.5) | 1 (1.9) |
| Chronic kidney disease | 40 (25.5) | 31 (57.4) |
| COPD | 21 (13.4) | 4 (7.4) |
| Asthma | 8 (5.1) | 1 (1.9) |
| Pulmonary hypertension | 19 (12.1) | 4 (7.4) |
| Moderate/severe AR | 17 (10.8) | 0 |
| Moderate mitral regurgitation | 16 (10.2) | 1 (1.9) |
| Moderate tricuspid regurgitation | 15 (9.6) | 2 (3.7) |
| STS Score (%) | 4.7±3.4 | 3.2 ± 2.0 |
| EuroScore II | 5.1±4.9 | 4.0 ± 3.2 |
| Haemoglobin, g/dL | 11.9±2.0 | 12.7 ± 1.5 |
| Serum creatinine, μmol/L | 1.2±1.1 | 1.1 ± 0.4 |
| Creatinine >2 mg/dL | 10 (6.4) | 3 (5.6) |
| eGFR, mL/min/1.73m2 | 59.0±21.6 | 62.01 ± 15.5 |
| LVEF, % | 56.4±13.2 | 54.6 ± 5.6 |
| Mean aortic gradient, mmHg | 49.5±18.5 | 53.4 ± 14.24 |
| Aortic valve area, cm2 | 0.7±0.2 | 0.68 ± 0.15 |
| Prior stroke | 13 (8.3) | 3 (5.6) |
| Prior TIA | 0 | 0 |
Values are n (%) or mean ± SD.
Procedural Characteristics
| Variables | One-year Follow-up Cohort (N=157) | Long-term Follow-up Cohort (N=54) |
|---|---|---|
| Trans-femoral Access | 157 (100) | 54 (100) |
| 18 Fr introducer sheath (Cook Check-Flo) | 157 (100) | 54 (100) |
| Local anaesthesia and conscious sedation | 69 (43.9) | 48 (88.9) |
| Implanted valve size | ||
| Hydra 22 mm | 4 (2.5) | 0 |
| Hydra 26 mm | 72 (45.9) | 19 (35.2) |
| Hydra 30 mm | 81 (51.6) | 35 (64.8) |
| Pre-dilatation | 126 (80.3) | 53 (98.1) |
| Post-dilatation | 81 (51.6) | 27 (50.0) |
| Procedure time, min | 122.8±57.0 | 116.8 ± 36.4 |
| Fluoroscopy time, min | 31.7±18.8 | 27.6 ± 12.0 |
| Contrast Volume, mL | 130.7±72.2 | 140.4 ± 50.3 |
| Duration of hospital stay, days | 7.2±6.3 | 8.8 ± 7.7 |
Values are n (%) or mean ± SD.
Device Success
| Variables | N=157 | 95% CI |
|---|---|---|
| Device Success | 130 (82.8%) | 75.8, 88.2 |
| Successful implantation | 148 (94.3%) | 89.0, 97.2 |
| One valve implanted (i.e. no need for second valve) | 148 (94.3%) | 89.0, 97.2 |
| Proper anatomical location | 146 (93.0%) | 87.5, 96.3 |
| Single valve in correct position | 142 (90.3%) | 84.5, 94.4 |
| Aortic valve area >1.2 cm2 | 145 (92.4%) | 86.7, 95.8 |
| Mean aortic valve gradient <20 mmHg | 150 (95.5%) | 90.7, 98.0 |
| Moderate or severe paravalvular leak | 11 (7.0%) | 3.3, 11.7 |
| Prosthesis–patient mismatch | 22 (14.0%) | 9.2, 20.7 |
| Severe prosthesis–patient mismatch | 9 (5.7% | 2.8, 11.0 |
| Procedural mortality | 3 (1.9%) | 0.2, 5.0 |
Values are n (%) or mean ± SD.